When you hear the terms “pH” and “alkalinity,” you might wonder if they mean the same thing or if one is more important than the other. Understanding the difference between pH vs alkalinity is key to keeping your water, pool, or even your garden healthy and balanced.
If you want to avoid costly mistakes and make smarter decisions about your water quality, this article is for you. By the end, you’ll know exactly what each term means, how they affect each other, and why both matter to you.
Let’s dive in and clear up the confusion once and for all.
Credit: blog.orendatech.com
What Ph Measures
Understanding what pH measures is essential for many areas, like gardening, cooking, and health. pH tells us how acidic or basic a liquid is. This helps us know if the liquid can react with other substances or if it is safe for use. The concept can seem tricky, but it’s simple once you know the basics.
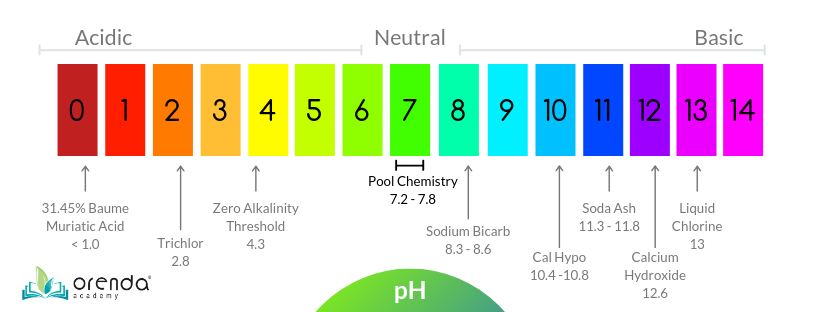
Acidity And Basicity Scale
The pH scale measures acidity and basicity. It runs from 0 to 14. Numbers below 7 show acidity. Numbers above 7 show basicity, also called alkalinity. A pH of exactly 7 means the liquid is neutral. Water usually has a neutral pH.
Ph Range Explained
The scale is logarithmic, so each number changes the acidity ten times. For example, pH 4 is ten times more acidic than pH 5. Low pH liquids have more hydrogen ions. High pH liquids have fewer hydrogen ions and more hydroxide ions. This balance defines how acidic or basic the liquid is.
Common Ph Examples
Many common liquids have known pH values. Lemon juice is very acidic, with a pH around 2. Baking soda dissolved in water is basic, with a pH near 9. Pure water is neutral, with a pH close to 7. Knowing these helps you understand pH better.
What Alkalinity Measures
Alkalinity measures how well water can resist changes in pH. It shows the water’s ability to neutralize acids. This is important for keeping water chemistry stable and safe for plants, animals, and people.
Buffering Capacity
Alkalinity acts like a buffer in water. It stops sudden shifts in pH levels. This helps maintain a balanced environment. Water with good buffering capacity stays healthy for longer.
Role In Water Chemistry
Alkalinity controls acid-base reactions in water. It affects how chemicals dissolve and react. This balance influences water quality and clarity. It also impacts the survival of aquatic life.
Sources Of Alkalinity
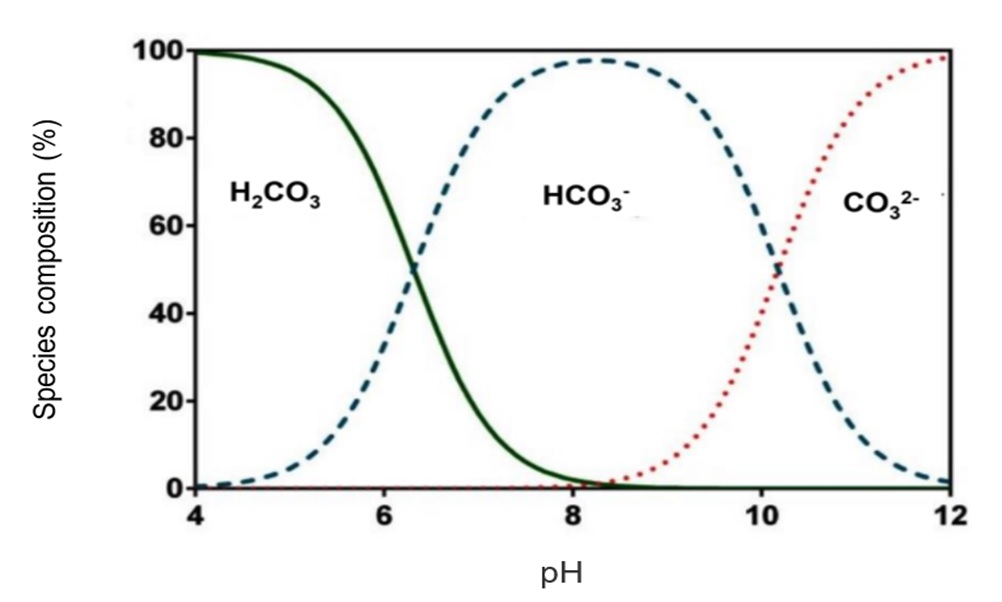
Alkalinity comes from minerals like bicarbonates, carbonates, and hydroxides. These minerals enter water from rocks and soil. Human activities can also add alkalinity through pollution or treatment chemicals.
How Ph And Alkalinity Interact
pH and alkalinity are closely connected in water chemistry. They affect each other’s levels and behavior. Understanding their interaction helps manage water quality in many settings.
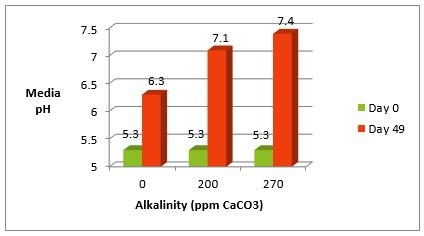
Alkalinity acts as a buffer that keeps pH levels steady. Without enough alkalinity, pH can change quickly. This can cause problems for aquatic life and water systems.
Ph Stability
Alkalinity controls how much acid or base water can handle. It prevents large swings in pH. Stable pH means water is less acidic or basic over time.
This stability is vital for maintaining safe water conditions. Low alkalinity means pH can drop or rise fast. Such changes stress living organisms and damage equipment.
Impact On Aquatic Life
Fish and other aquatic animals need stable pH to survive. Sudden pH changes can harm their gills and skin. Alkalinity keeps pH in a safe range for these creatures.
Low alkalinity water often leads to acidic conditions. Acidic water can dissolve important minerals and release toxins. Stable alkalinity protects aquatic ecosystems from harm.
Effects On Water Treatment
Water treatment depends on balanced pH and alkalinity. Alkalinity helps control pH during chemical processes. This ensures treatments like chlorination work properly.
Without proper alkalinity, pH can swing and reduce treatment efficiency. It may cause pipe corrosion or scale buildup. Managing both pH and alkalinity is key for safe water supply.
Credit: www.pthorticulture.com
Testing Ph And Alkalinity
Testing pH and alkalinity is essential for many water-related tasks. It helps keep water safe and balanced. Knowing both values guides you to correct problems early. This avoids damage to pipes, plants, or pools. Testing is easy with the right tools and steps.
Measurement Methods
pH is tested using strips, meters, or drops. Strips change color quickly and are simple to use. Meters give digital readings and are very accurate. Drop tests use chemical reagents and show colors to compare.
Alkalinity tests often use liquid kits with drops. Adding drops to water causes color change. Count the drops until color shifts to find alkalinity levels. Some kits combine pH and alkalinity tests in one.
Interpreting Test Results
pH shows how acidic or basic water is. A pH of 7 is neutral. Below 7 means acidic, above means alkaline. Alkalinity measures water’s ability to resist pH change. Higher alkalinity means more stability.
Ideal pH and alkalinity levels depend on the use. Pools usually need pH between 7.2 and 7.8. Alkalinity should stay within 80 to 120 ppm. Test results help decide if chemicals are needed.
Common Testing Mistakes
Not testing regularly can miss important changes. Using expired test kits gives wrong results. Not rinsing test tools before use causes errors. Testing water at wrong temperatures affects accuracy.
Mixing samples from different spots hides true values. Reading colors in poor light leads to mistakes. Testing without following instructions causes confusion. These errors can waste time and money.
Adjusting Ph And Alkalinity
Adjusting pH and alkalinity is key to keeping water balanced. Both affect water quality and health. Proper adjustment prevents damage to pipes and fixtures. It also keeps water safe for use.
Chemical Treatments
Chemicals can quickly change pH and alkalinity levels. Acidic chemicals lower pH and reduce alkalinity. Common acids used include muriatic acid and sodium bisulfate. To raise pH and alkalinity, add baking soda or soda ash. Use chemicals carefully and follow instructions. Overuse can cause harmful water conditions.
Natural Adjustment Methods
Natural methods adjust water without harsh chemicals. Adding crushed limestone can slowly raise alkalinity. Aeration helps reduce carbon dioxide, raising pH naturally. Using plants or certain minerals can also help balance water. These methods work slower but are safer for the environment.
Monitoring Changes
Regular testing is vital after adjustments. Use pH and alkalinity test kits for accuracy. Test water at different times to track changes. Record results and adjust treatments as needed. Consistent monitoring keeps water balanced and healthy.
Practical Applications
Understanding pH and alkalinity is important in many real-life situations. Both affect water quality and chemical balance. Knowing how to manage them helps keep systems safe and efficient. Below are key areas where pH and alkalinity play a big role.
Swimming Pool Maintenance
Proper pH and alkalinity keep pool water clean and safe. pH affects swimmer comfort and sanitizer effectiveness. Alkalinity acts like a buffer to stabilize pH levels. Low alkalinity causes pH to change quickly. High alkalinity can make water cloudy. Balancing both prevents corrosion and scaling on pool surfaces. Regular testing helps maintain the right balance.
Aquarium Care
Fish and plants need stable pH and alkalinity to thrive. pH affects fish health and biological functions. Alkalinity helps keep pH steady despite waste and feeding. Sudden pH shifts stress aquatic life and cause illness. Aquarium owners use test kits to monitor both values. Adjusting water chemistry supports a healthy underwater environment.
Agricultural Uses
Soil pH and alkalinity influence crop growth and nutrient uptake. Certain plants prefer acidic or alkaline soils. Proper pH ensures nutrients are available to roots. Alkalinity affects soil’s ability to resist acid rain and fertilizers. Farmers test soil regularly to manage pH and alkalinity levels. Correct balance improves yield and soil health over time.
Health And Environmental Impacts
Understanding the health and environmental impacts of pH and alkalinity is important. These factors affect water quality and safety. Both pH and alkalinity influence how water interacts with the body and the environment. Changes in either can lead to serious problems.
Effects On Drinking Water
Water with very low or high pH can harm health. Acidic water may corrode pipes, adding metals like lead. High pH water can taste bitter and cause skin irritation. Alkalinity helps buffer water, keeping pH stable. Low alkalinity means water pH can change quickly, risking safety. Safe drinking water requires balanced pH and alkalinity levels.
Environmental Concerns
Water pH affects aquatic life survival. Most fish and plants need pH between 6.5 and 9. Extreme pH damages gills and disrupts reproduction. Alkalinity protects ecosystems by reducing pH swings. Acid rain lowers pH in lakes, harming wildlife. Maintaining proper alkalinity helps ecosystems resist acid changes. Pollutants often alter pH and alkalinity, causing environmental stress.
Regulatory Standards
Governments set rules to control pH and alkalinity in water. Safe pH levels usually range from 6.5 to 8.5 for drinking water. Alkalinity standards vary but support pH balance. Water suppliers must monitor and adjust these levels regularly. Regulations help prevent health risks and environmental damage. Compliance ensures water stays safe for people and nature.
Credit: cropaia.com
Frequently Asked Questions
What Is The Difference Between Ph And Alkalinity?
PH measures how acidic or basic water is on a scale of 0 to 14. Alkalinity shows water’s ability to neutralize acids, acting as a buffer. Both affect water quality but measure different properties.
How Does Alkalinity Affect Ph Levels?
Alkalinity helps keep pH stable by neutralizing acids in water. Higher alkalinity means pH changes slowly and stays balanced. Low alkalinity can cause pH to change quickly and harm water systems.
Why Is Measuring Ph Important In Water Testing?
PH affects chemical reactions, water taste, and safety for plants and animals. Testing pH helps identify pollution or contamination in water. It ensures water is safe for drinking and use.
Can High Alkalinity Harm Aquatic Life?
Too much alkalinity can raise pH to unsafe levels for fish and plants. It may cause stress or damage to aquatic life over time. Balanced alkalinity supports a healthy environment.
How Do You Adjust Ph And Alkalinity In Water?
To raise pH, add alkaline substances like baking soda or lime. To lower pH, use acidic substances like vinegar or muriatic acid. Adjusting alkalinity usually involves adding baking soda to increase it.
Conclusion
PH and alkalinity both affect water quality but in different ways. PH measures how acidic or basic the water is. Alkalinity shows water’s ability to neutralize acids. Knowing both helps keep water balanced and safe. Test them regularly to avoid problems.
Clear water depends on managing pH and alkalinity well. Small changes can make a big difference. Stay informed and take simple steps to maintain good water health.
